What is Ductility?
The ductility of a material is its ability to permanently deform in response to stress (e.g., stretch, bend, or spread). For example, the majority of common steels are highly ductile and resistant to localized stress concentrations.
Due to their lack of ductility, brittle materials such as glass cannot withstand stress concentrations and break easily. Under pressure, a material specimen initially deforms elastically, but once it reaches a certain deformation (known as the elastic limit), the deformation becomes permanent.
In materials research, ductility is the degree to which a material can withstand plastic deformation under tensile stress before failing.
In engineering and manufacturing, ductility is essential for determining a material’s suitability for particular manufacturing processes (such as cold working) and its resistance to mechanical stress. Gold and copper are typical examples of ductile substances.
The malleability of a material, a related mechanical property, is determined by its ability to deform plastically without breaking under compressive force. In the past, materials were considered malleable if they could be hammered or rolled into desired shapes. Lead is a material that is somewhat malleable but not ductile.
Examples
The majority of metals, including gold, silver, copper, erbium, terbium, samarium, aluminum, and steel, are examples of ductile materials. The metals tungsten and high-carbon steel are examples of non-ductile materials. Generally speaking, nonmetals are not ductile.
How to Measure Ductility
The ductility of a metal is its ability to undergo deformation without cracking. Metals that are ductile are capable of being pushed or shaped without cracking. Metals that are brittle can break (essentially the opposite of ductile).
Formability is substantially affected by ductility. Extremely brittle metals may not be able to be manufactured satisfactorily. For instance, a metal must possess a certain degree of ductility in order to be stretched into a thin wire.
If the metal is too brittle, it will shatter as soon as it begins to expand. Ductility is another essential safety factor for structural formations. When structures are subjected to heavy stresses, ductility allows them to bend and deform without breaking.
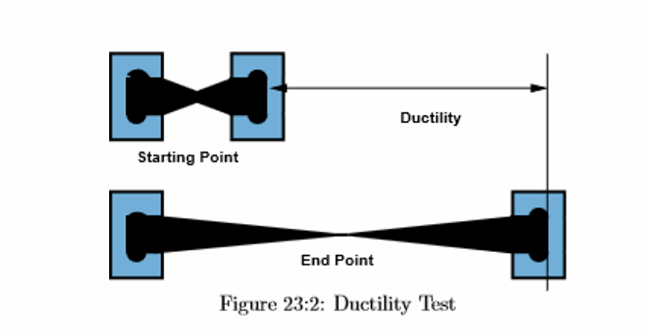
There are two ways to evaluate ductility: percentage elongation and percentage reduction.
During a tensile test, when a metal is dragged until it breaks, its length deforms as a percentage of its original length and is measured as percentage elongation.
The percentage decrease of a specimen’s narrowest cross-section after a tensile test-induced rupture.
The temperature has an effect on plasticity. Consequently, it is crucial to consider the temperatures the metal will encounter in a given application. A chart depicting the transition temperature between ductile and brittle for the majority of metals is available and valuable.
Which Metals Are Ductile?
Aluminum, brass, copper, low-carbon steel, gold, silver, tin, and lead are examples of ductile metals. The metals wrought iron, chromium, and tungsten are examples of brittle materials. Cables, stampings, and structural beams are examples of applications that require high ductility.
Materials Science
Gold is exceptionally malleable. It can be stretched until it breaks and transforms into a single-strand wire.
Under stress, it is impossible to manipulate materials using metal-forming operations such as hammering, rolling, drawing, or extrusion with materials that crack, break or shatter. Materials that are brittle can be cast or thermoformed, whereas materials that are malleable can be formed by stamping or pressing.
Metallic bonds, which are predominant in metals and confer high degrees of ductility, lend metals their reputation for being generally ductile. In metallic bonds, valence shell electrons are delocalized and shared by multiple atoms.
Delocalized electrons allow metal atoms to slide past one another, preventing other materials from shattering due to strong repulsive interactions.
The ductility of steel varies depending on the alloying elements. As carbon content increases, ductility decreases. Many amorphous solids and polymers, such as Play-Doh, can be deformed. Gold is the most ductile metal, while platinum is the most malleable.
Through the formation, reorientation, and migration of dislocations and crystal twins, such metals deform under intense stretching without appreciable hardening.
Factors That Affect the Ductility of Metals:
Both internal and external factors, such as composition, grain size, cell structure, etc., and hydrostatic pressure, temperature, previously experienced plastic deformation, etc., influence ductility.
Below are a few crucial points regarding ductility:
Compared to metals with HCP crystal structures, those with FCC and BCC crystal structures exhibit superior high-temperature ductility.
The grain size has a significant effect on ductility. On a scale of a few microns, many alloys exhibit superplastic behavior when the grain size is excellent.
Higher oxygen content decreases the ductility of steel. Even relatively low levels of impurities have a significant impact on the ductility of some alloys. At approximately 1040°C, the ductility of carbon steels containing sulphur impurities as small as 0.018% decreases rapidly. However, if the Mn concentration is high, this can be corrected. At 1040 °C, the Mn/S ratio is what determines the ductility of carbon steels. When this ratio is 2, the elongation percentage at 1040 °C is only 12-15%, but when it is 14, it is 110 percent.
The temperature has a substantial effect on ductility and, by extension, formability. It promotes ductility in general, but at certain temperatures, it may decrease due to phase transition and microstructural changes induced by temperature rise. Temperature influences the ductility of stainless steel.
At 1050 °C, it is least ductile, and at 1350 °C, it is most ductile. Consequently, its hot operating range is quite constrained.
Hydrostatic pressure improves the ductility of a material. In torsion testing, the length of the specimen decreases as the strength of the torsion increases. In the torsion test, the specimen demonstrates greater ductility when subjected to axial compressive stress than when none is applied. The application of axial tension further diminishes ductility.
Read More :
Learn About The Applications Of Alloy 20 Pipes: Alloy 20 is one of the “Super” stainless steels with the highest acid attack resistance. The presence of nickel, chromium, molybdenum, and copper increases the material’s resistance to chloride stress corrosion, which can lead to cracking and general pitting. Columbium is added to the alloy to prevent carbide precipitation during welding.
Selecting Metals and Alloys for Medical Device Manufacturing: The work quality of metal component manufacturers who supply the medical device industry has recently improved significantly. The product quality of these alloys has never been better since the implementation of statistical process control, ISO 9000 certifications, and other initiatives.